|
9/11/2023 0 Comments Defects in crystal lattice 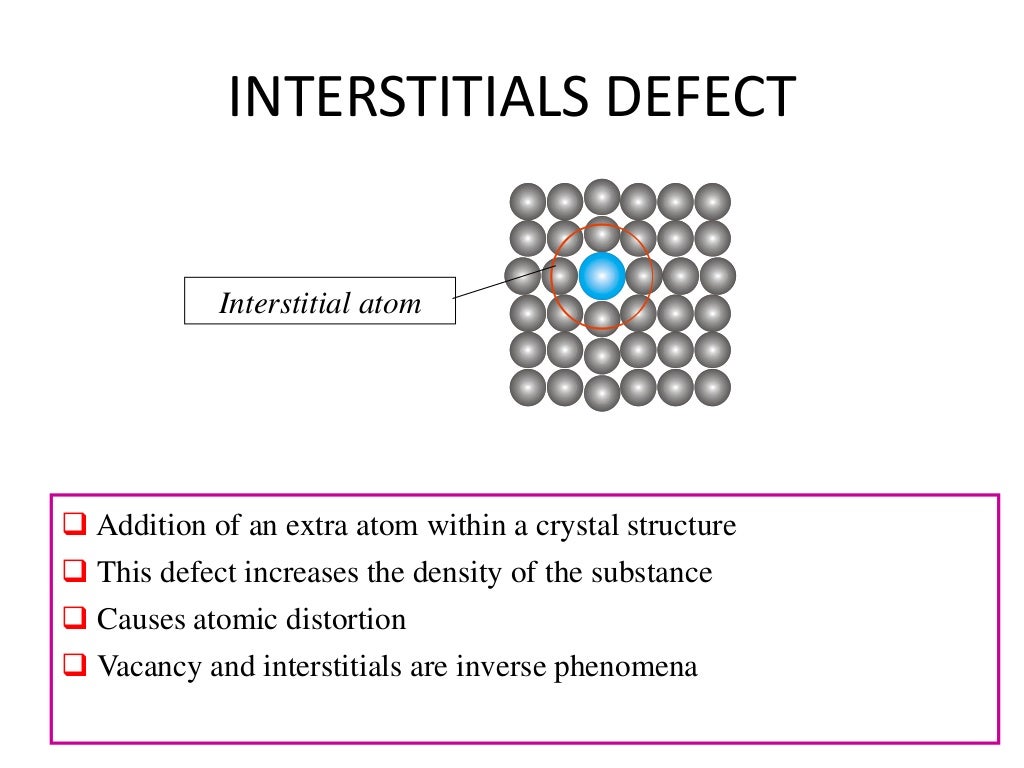
Electron diffraction and surface probe microscopies have resolved ordered cation vacancies and surface reconstructions in magnetite 26, 27, 29, 30, 31. 
X-ray diffraction and Mössbauer spectroscopy have been widely used to measure the average unit-cell parameters and stoichiometry of magnetite powders 17, 19. Structural characterization of magnetite crystals has been conducted using various experimental methods 8, 16, 17, 26, 27, 28. Both reductive growth and oxidative dissolution reactions are controlled by diffusion of Fe(II) in and out of the spinel lattice 25. In particular, off-stoichiometric magnetite can undergo a reverse reaction, i.e., reductive growth in the presence of aqueous Fe(II) 24. Strain and defects in minerals are important factors in controlling kinetics of geochemical reactions, such as mineral dissolution/growth and isotope exchange reactions 21, 22, 23. Since there is a continuous solid solution between magnetite and maghemite, removal of Fe(II) from magnetite leads to the formation of off-stoichiometric magnetite (Fe(II)/Fe(III) ratio between 0.5 and 0) and lattice contraction 19, 20, but three-dimensional (3D) observations of the lattice spacing variation with associated strain and defect structures remain limited 16, 17. ( 1) represented the oxidative dissolution of magnetite, where Fe(II) and Fe(III) in different sites are denoted by T (tetrahedral) and O (octahedral) with □ representing the cation vacancies. However, there is limited experimental evidence to test this structure because the crystal structures of magnetite and maghemite are nearly identical: their lattice constants differ by ~1%, and the spinel lattice is preserved, while the extra charge left by Fe (II) vacancies is neutralized by oxidation of Fe(II) to Fe(III) in maghemite 19, 20. This is consistent with a core–shell structure model where a partially oxidized magnetite crystal has a magnetite core and a maghemite-like shell 18. Observed oxidation kinetics in both dissolution and heated magnetite crystals are in agreement with a spherical diffusion model involving outward diffusion of Fe(II) to the mineral surfaces 15, 16, 17. Several issues regarding magnetite oxidation mechanisms remain unresolved. Understanding such a phenomenon can provide insights into controlling the reactivity of magnetite used for groundwater remediation and can benefit the proper interpretation of magnetic fields recorded in its domain structure when the mineral was formed (i.e., a paleomagnetic proxy) 12, 13, 14. 
Both oxidative dissolution and oxidation with increasing O 2 fugacity and temperature lead to a decrease in the Fe(II)/Fe(III) ratio from 0.5 in stoichiometric magnetite to 0 in maghemite (γ-Fe 2O 3) 11. Oxidative dissolution of magnetite releases Fe(II), a critical reductant for immobilizing heavy metals and radionuclides in subsurface environment 9, 10. In aqueous environments, magnetite plays an important role as a recyclable geochemical battery, where electrons are stored/released in the redox active Fe(II)/Fe(III) couple that serves as the main energy reservoir for Fe-metabolizing bacteria 1, 8. Magnetite is commonly observed in igneous and metamorphic rocks on Earth and has been found in meteorites and rocks on Mars, and it can also be formed through biomineralization processes 5, 6, 7. Magnetite (Fe 3O 4) is an iron oxide mineral with an inverse spinel-type structure containing both Fe(II) and Fe(III) 4. Redox cycling of iron (Fe) is an essential chemical process in geo- and bio-spheres 1, 2, 3. These results demonstrate that oxidative dissolution of magnetite can induce a rich array of strain and defect structures, which could be an important factor that contributes to the high reactivity observed on magnetite particles in aqueous environment. In contrast, strain evolution is less pronounced during magnetite oxidation at elevated temperature in air. This heterogeneous strain likely results from lattice distortion caused by Fe(II) diffusion that leads to the observed domains of increasing compressive and tensile strains. Oxidative dissolution in acidic solutions leads to increases in the magnitude and heterogeneity of internal strains. Here we show that the morphology and internal strain distributions within individual nano-sized (~400 nm) magnetite crystals can be visualized using Bragg coherent diffractive imaging (BCDI). Spatially resolving strain fields and defect evolution during oxidation of magnetite provides further insight into its reaction mechanisms. Oxidation of magnetite (Fe 3O 4) has broad implications in geochemistry, environmental science and materials science. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
